Naphtha
| Infobox on Naphtha | |
|---|---|
| Example of Naphtha | 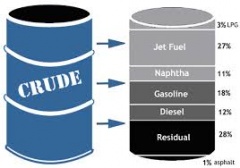 |
| Facts | |
| Origin | - |
| Stowage factor (in m3/t) | - |
| Humidity / moisture | - |
| Ventilation | - |
| Risk factors | See text |
Naphtha
Description / Application
Naphtha is obtained in petroleum refineries as one of the intermediate products from the distillation of crude oil. It is a liquid intermediate between the light gases in the crude oil and the heavier liquid kerosene. Naphthas are volatile, flammable and have a specific gravity of about 0.7. The generic name 'naphtha' describes a range of different refinery intermediate products used in different applications. Naphtha is a deep amber to dark red liquid, a mixture of xylene and higher homologs; crude, dark straw-coloured liquid; refined, water-white liquid.
Naphtha normally refers to a number of different flammable liquid mixtures of hydrocarbons, i.e. a component of natural gas condensate or a distillation product from petroleum or coal tar boiling in a certain range and containing certain hydrocarbons. It is a broad term covering among the lightest and most volatile fractions of the liquid hydrocarbons in petroleum. Naphtha is a colourless to reddish-brown volatile aromatic liquid, very similar to gasoline.
In petroleum engineering, full range naphtha is defined as the fraction of hydrocarbons in petroleum boiling between 30°C and 200°C. It consists of a complex mixture of hydrocarbon molecules generally having between 5 and 12 carbon atoms. It typically constitutes 15–30% of crude oil, by weight. Light naphtha is the fraction boiling between 30 °C and 90 °C and consists of molecules with 5–6 carbon atoms. Heavy naphtha boils between 90°C and 200°C and consists of molecules with 6–12 carbons.
Naphtha is used primarily as feedstock for producing high octane gasoline (via the catalytic reforming process). It is also used in the bitumen mining industry as a diluent, the petrochemical industry for producing olefins in steam crackers, and the chemical industry for solvent (cleaning) applications. Common products made with it include lighter fluid, fuel for camp stoves, and some cleaning solvents.
Petroleum naphtha is an intermediate hydrocarbon liquid stream derived from the refining of crude oil. It is most usually desulfurized and then catalytically reformed, which re-arranges or re-structures the hydrocarbon molecules in the naphtha as well as breaking some of the molecules into smaller molecules to produce a high-octane component of gasoline (or petrol).
There are hundreds of different petroleum crude oil sources worldwide and each crude oil has its own unique composition or assay. There are also hundreds of petroleum refineries worldwide and each of them is designed to process either a specific crude oil or specific types of crude oils. That means that it is virtually impossible to provide a definitive, single definition of the word naphtha since each refinery produces its own naphthas with their own unique initial and final boiling points and other physical and compositional characteristics. In other words, naphtha is a generic term rather than a specific term.
In addition, naphthas may also be produced from coal tar, shale deposits, tar sands such as in Canada, the destructive distillation of wood and coal gasification or biomass gasification to produce a syngas followed by the Fischer-Tropsch process to convert the syngas into liquid hydrocarbon products. For that reason, this article is entitled Petroleum naphtha and deals only with naphthas produced by the processing of crude oil in petroleum refineries.
The first unit process in a petroleum refinery is the crude oil distillation unit. The overhead liquid distillate from that unit is called virgin or straight-run naphtha and that distillate is the largest source of naphtha in most petroleum refineries. The naphtha is a mixture of very many different hydrocarbon compounds. It has an initial boiling point (IFP) of about 35°C and a final boiling point (FBP) of about 200°C, and it contains paraffin, naphthene (cyclic paraffins) and aromatic hydrocarbons ranging from those containing 4 carbon atoms to those containing about 10 or 11 carbon atoms.
The virgin naphtha is often further distilled into two streams:
- a virgin light naphtha with an IFP of about 30°C and a FBP of about 145°C containing most (but not all) of the hydrocarbons with 6 or less carbon atoms
- a virgin heavy naphtha containing most (but not all) of the hydrocarbons with more than 6 carbon atoms. The heavy naphtha has an IFP of about 140 °C and a FBP of about 205°C.
It is the virgin heavy naphtha that is usually processed in a catalytic reformer because the light naphtha has molecules with 6 or less carbon atoms which, when reformed, tend to crack into butane and lower molecular weight hydrocarbons which are not useful as high-octane gasoline blending components. Also, the molecules with 6 carbon atoms tend to form aromatics which is undesirable because governmental environmental regulations in a number of countries limit the amount of aromatics (most particularly benzene) that gasoline may contain.
Some refinery naphthas also contain some olefinic hydrocarbons, such as naphthas derived from the fluid catalytic cracking, visbreakers and coking processes used in many refineries. Those olefin-containing naphthas are often referred to as cracked naphthas.
In some (but not all) petroleum refineries, the cracked naphthas are desulfurized and catalytically reformed (as are the virgin naphthas) to produce additional high-octane gasoline components.
Some petroleum refineries also produce small amounts of specialty naphthas for use as solvents, cleaning fluids, paint and varnish diluents, asphalt diluents, rubber industry solvents, dry-cleaning, cigarette lighters, and portable camping stove and lantern fuels. Those specialty naphthas are subjected to various purification processes.
Sometimes the specialty naphthas are called petroleum ether, petroleum spirits, mineral spirits, paraffin, benzine, hexanes, ligroin, white oil or white gas, painters naphtha, refined solvent naphtha and Varnish makers' & painters' naphtha (VM&P). The best way to determine the boiling range and other compositional characteristics of any of the specialty naphthas is to read the Material Safety Data Sheet (MSDS) for the specific naphtha of interest.
On a much larger scale, petroleum naphtha is also used in the petrochemicals industry as feedstock to steam reformers and steam crackers for the production of hydrogen (which may be and is converted into ammonia for fertilizers), ethylene and other olefins. Natural gas is also used as feedstock to steam reformers and steam crackers.
Shipment / Storage / Risk factors
Flammable, dangerous fire risk, explosive limits in air 1-6%. Toxic by ingestion, inhalation and skin absorption.
Forms of naphtha may be carcinogenic, and frequently products sold as naphtha contain some impurities which may also have harmful properties of their own. Like many hydrocarbon products, they are products of a refining process in which a complex soup of chemicals is broken into another range of chemicals, which are then graded and isolated mainly by their specific gravity and volatility. There is, therefore, a range of distinct chemicals included in each product. This makes rigorous comparisons and identification of specific carcinogens difficult, especially in our modern environment where people are daily exposed to many such products, and is further complicated by exposure to a significant range of other known and potential carcinogens.
Light naphtha is a mixture consisting mainly of straight-chained and cyclic aliphatic hydrocarbons having from five to nine carbon atoms per molecule. Heavy naphtha, a mixture consisting mainly of straight-chained and cyclic aliphatic hydrocarbons having from seven to nine carbons per molecule. Almost all volatile, lipid-soluble organic chemicals cause general, nonspecific depression of the central nervous system or general anesthesia. The OSHA PEL TWA = 100 parts-per-million (ppm); Health Hazards/Target Organs = eyes, skin, RS, CNS, liver, kidney. Symptoms of acute exposure are dizziness and narcosis with loss of consciousness. The World Health Organization categorizes health effects into three groups: reversible symptoms (Type 1), mild chronic encephalopathy (Type 2) and severe chronic toxic encephalopathy (Type 3).
Topical exposure to naphtha can cause a burning sensation on the skin within a period of minutes to an hour, followed by contact dermatitis—a rash—that can last for days to weeks.
Below are linked few Material Safety Data Sheet (MSDS) specifications for different "naphtha" products containing varying degrees of naphtha, as well as various other chemicals. As well as giving health guidelines, these are some of the few ways to determine what a given product contains.
- JT Baker VM&P Naphtha MSDS
- Diggers Shellite MSDS
- Shell Ronsonol MSDS source1 formula developed for Ronson
- Links to more MSDS for various camping-stove fuels including several that include naphtha
Benzene in particular is a known high-risk carcinogen, so benzene content is typically specified in the MSDS when it is present in the mixture due to the specifics of the feedstock and distilling process used.
Naphtha is also extremely volatile and can explode on exposure to high temperature surfaces.
Note:
For overseas carriage aspects of Chemicals, the readers are recommended to acquire or have access to a good chemical dictionary, and a copy of the International Maritime Dangerous Goods (IMDG) Code, issued by the International Maritime Organisation. Also consult the applicable MSDS sheet.











